Results & impact 3 February 2026
- Home
- Press area
- Press releases
- Tackling antibiotic resistance
Towards new immunotherapies to tackle antibiotic resistance
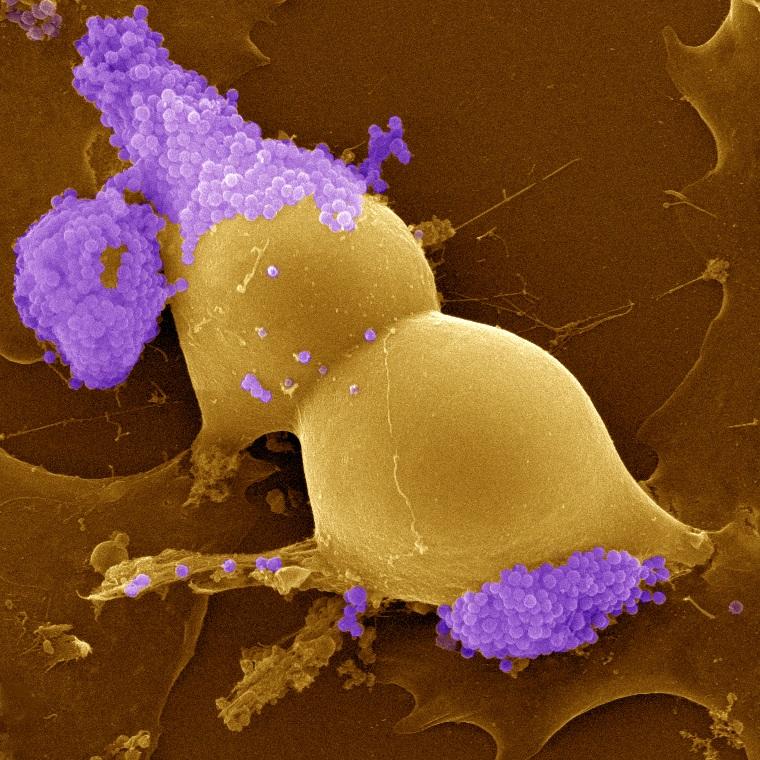
Ehrlichia ruminantium bacteria (purple) infecting a bovine aortic endothelial cell in the process of division © D. Meyer, CIRAD
Determining how bacteria attack and with which proteins, in order to develop suitable shields for the cells targeted: this is the aim of the research published on 12 July 2021 in PLOS Computational Biology by two researchers from CIRAD. They have identified virulence factors for the Anaplasmataceae family of bacteria that includes human pathogens. Ehrlichia ruminantium, a representative of this family, is responsible for heartwater, a fatal disease in ruminants that causes heavy losses for livestock farms in Sub-Saharan Africa and islands of the Indian Ocean and the Caribbean.
Central to this research is the fight against bacterial resistance to antibiotics. Damien Meyer, a researcher at CIRAD and co-author of the study, explains: “In 2019, WHO reported that antibiotic resistance could claim as many as 10 million victims per year by 2050. The cause is the overuse of antibiotics, especially in the agricultural sector, to prevent disease outbreaks in livestock farms. Our objective is to develop alternatives to antibiotics, by focusing on targeted immune strengthening strategies”.
The first stage consists in finding out how bacteria attack: “what are the cellular pathways that they hijack?”.
By improving understanding of bacterial virulence factors, the scientists can identify the weak points of the cells attacked. This enables them to develop targeted defences to strengthen these weaknesses.
“The bacterium still attacks, but now fires blanks”
“The idea is simply to provide better shields for cells”, continues Damien Meyer. “Rather than trying to destroy bacteria with antibiotics, we let them attack, except they are now firing blanks”.
New therapies, directed toward the host rather than the pathogen, are currently being studied.
Damien Meyer insists on the urgent need to develop alternatives to antibiotics: “By working on ways to strengthen the immunity of host cells, we avoid the selection pressure exerted by the use of antibiotics. This pressure results in the mutation of certain bacteria, which then become resistant to antibiotics. If we continue on this path, some diseases that are relatively “common” today could become untreatable in a few years’ time”.
The virulence proteins were identified using the S4TE software package, also developed by CIRAD.
An international patent to advance vaccination against heartwater
After 10 years of research and administrative processes, the research team from CIRAD, based in Guadeloupe, has also obtained an international patent for the discovery of a virulence attenuator gene in Ehrlichia ruminantium, the bacterium that causes heartwater. This major advance paves the way for the development of vaccines through the generation of attenuated bacterial strains.
With this international patent, CIRAD ensures it will retain the rights to this discovery in order to guarantee the accessibility of vaccines for the poorest countries. The development of vaccines, as an alternative strategy to antibiotic use, is also part of the fight against antibiotic resistance.
Reference
Christophe Noroy,Damien F. Meyer. 2021. The super repertoire of type IV effectors in the pangenome of Ehrlichia spp. provides insights into host-specificity and pathogenesis. PLOS Computational Biology.